COMPULSORY TO SING ALONG !
Wednesday, September 17, 2008
Thursday, September 4, 2008
non- concerning chpt789
He said:''Magic has so much towards science..the reaon that I do this show is because science is fun & worth studying..Without science, the world would not function ..You will not have popcorn..gameboy.." SO MEANINGFUL !
He also teaches a trick that you can do at home too!!!
:)
Wednesday, September 3, 2008
Helpless
Tuesday, September 2, 2008
Last but not least, chpt 9

What is internal energy?
Okay, internal energy is simply Kinetic & potential energy.
=You know how to remember?? haha..
I am a K-Poh (Oh no! Im insulting myself..haha, see, Im sacrificing myself for you!)
Kinetic energy is due the vibration of the particles which closely relates to temperature while Potential energy is due the stretching and compressing of molecular bonds
Hence, an increase of temperature would increse kinetic component

Melt, Melt, Melt !
Melting is basically the change of state from solid to liquid.
Imp**During melting, the temperature remains constant at the melting point where thermal energy is absorbed by the substance
As the solids are in fixed position, therefore, the intermolecules bonds must be broken for molecules to move
FREEZE, FREEZE, FREEZE !

Ok, give me a break! not this freeze...but in terms of states..
DO YOU KNOW ? WORLD ICE HOTEL.. no joke





Everything in the Ice Hotel is created from Mother Nature’s glory, from chairs carved out of snow, right down to drinking glasses made of ice. Go http://www.travelscribes.com/ice_hotel.htm for more details if you are intersted
Ok, thats just the intro of how ice can be so beautiful...
Freezing, is the change of state from liquid to solid, without change in temperature.
WHY ? temperature change occur only when substance is cooled within same state when liquid state(90 to 79 degree celcius)
Freezing point is 0 degree celcius..defintely, the pictures above have lower temperatures then 0 degree celcius.. or the hotel would become a swimming pool in no time! I wonder how cold that place will be..
Conversely, liquid molecules come together to solidify & intermolecules would form.
TRY IT OUT (This experiment was in the TB .. You can see the results straight away !
ok..it seems like Im advertising a slimming pill..diao..)
take note at 1:39 is quite funny !
Boiling
Boiling is the change of state from a liquid into vapour, occuring at constant temperature, boiling point
A
 Pic A shows how a normal boiling process.
Pic A shows how a normal boiling process. During this process, the temperature remains constant at its boiling point where thermal energy is absorbed.
B

Thermal energy would be used to seperate the molecules
C
 The molecules will start to seperate & move further apart..一刀两断
The molecules will start to seperate & move further apart..一刀两断 Condensation & evaporation
this cartoon video is quite interesting.. it educates you about condensation & evaporation.Let this be the intro of C&E.Happy watching!
Condensation

When does it occur?
When vapour changes into liquid at the same constant temperature. During this process, heat would be given out.
Similar to boiling, the temperature remains constant at the condensation point.
Evaporation

- When you simply blow your wet hands, evaporation would had already occur. The unique point of evaporation is that it can take place in any temperature. It would dries up any liquid, take for instance, even a swimming pool. But that may take months and it would be a SLOW PROCESS.[depending the variables that would speed up or slow down the evaporation process(will further elaborate below )]. The temperature may also change due to evaporation .
WHY WOULD WE FEEL COLD ?

Lets take for instance when we just came out of swimming pool feeling very cold..This is a common sceneria that would occur in ou common lifes, but have we ever wondered why isit cold? Is it just solely just the wind blowing against us ? As water evaporates, it takes away thermal energy from our skin. As a result, making us feel cold
From the video which educates you about C & E has already explained how evaporation occur. I will now explain again :D As the liquid surface, the ether molecules(more energetic) would overcome the downward attractive force of other ether molecules & escape to the atmosphere.
Evaporation is applied in our common lives. For e.g,
- the fridge that we use,
- to dry our wet clothes,
- to keep a person with high fever's temperature down
VIARABLES THAT AFFECT RATE OF EVAPORATION
Temperature- The higher the temperature of liquid, the increase rate of evaporation
Humidity in surrounding air- Rate of evaporation increase with decreasing humid.
Surface area-The higher the surface area, the faster the rate of evaporation.
Movement of air - The rate evaporation increases when the surrounding air is moving.
Pressure- Reducing atmospheric pressureincreases rate of evaporation.
Boiling point of liquid- Liquids with lower boiling point will increase the rate of evaporation
hope you learn something from my blog ;D thanks for reading!
----------------------------------end-----------------------------------
with my lovely pink
Jus a motivation to ASK ! (this applies to me too!)
Monday, September 1, 2008
chpt 8B , 8C, 8D, 8E :)

8 B ) Whats conduction ?
Its simply : transfer of thermal energy through a medium, without the medium moving

DO NOT------->
Conduction occurs when the vibration energy of molecules is transferred from 1 molecule to another.
When comparing metal and non-metal, metals have free electrons that make metal a good conductor of heat. So this explains why good conductor are capable of transferring thermal energy much faster than insulators.
-----------------------------------------------
kk..now after reading what I write, -->metals are capable of transferring thermal energy much faster than insulators, I guess you are capable enough to answer the video question below.
GO CHECK IT OUT !
GOT IT RIGHT ?
- How about the conduction in liquid ? Are they the same?
ANS: Obviously not ! ! haha. As the particles in liquid are further apart compared to solid
Lets check out this video which is somewhat similar to the TB experiment on pg 133(can refer while watching, this makes you understand better..muhahaha)
_______________________________________________
8 C )
----Whats ----
convection
?????ha???ha??????

Frankly speaking, I did not understand what the TB was saying until I see this video and can visualise better
- Convectional is the transfer of heat energy involving material of GAS OR LIQUID ONLY.
CONVECTION PROCESS
- Air above candle gets heated and expands
- This cause air to become less dense
- Then,air would rise
- Replaced with cool air at the end
_______________________________________________
8 D ) Whats radiation ?
radiation is the emission of infrared waves from surface of all bodies without using medium
Therefore, radiation CAN take place in a vacuum ( RMB? electromagnet waves chapt has stated in 1 of 8 of the properties that they do not require medium)
- good absorbers have dull and black surfaces while poor absorbers have shiny and white surfaces
FACTORS that affect infrared absorption
- SURFACE TEMPERATURE
Higher surface temperature gives higher rate of infrared radiation
- SURFACE AREA
Larger surface area gives higher emission of infrared radiation
- COLOUR & TEXTURE
Guess what ? If you don't know this, give a knock on your head..!!
I mention earlier ! The good absorbers must have dull & black surfaces. Therefore, black surface are better emitters .
8 E ) -----APPILCATIONS-----
of thermal energy transfer
Using what we learn in 8B-CONDUCTION-good conductors & insulators
GOOD CONDUCTORS (useful for thermal energy transferring quickly) are used in
- saucepan where direct heat is involved
- Heat exchanger to help save energy by using copper tubes
INSULATORS (to minimise loss of thermal energy)are used in
- irons, testube holder in our science lab too !
- sawdust to cover ice blocks
- wollen clothes keeps us warm!
CAN YOU THINK OF OTHER EXAMPLES IN OUR COMMON LIFE ?..there are so many things in the world that we take it for granted
 Using what we learn in 8C- CONVECTION
Using what we learn in 8C- CONVECTION
(its exactly what we learn in 8C)
*electric kettles - The bottom heating coil is placed bottom to aid transfer of thermal energy in water by convection.
- The heating coil heats up, expands & become less dense
- The heated water then rise
- The cooler upper part descend to replace the heated water.
*Refrigerator works just as the same way ;D
_______________________________________________
Using what we learn in 8D- RADIATION
teapots-  As mentioned, the shinier the surface, the worse emitter radiation it will be. Therefore, the shiny teapots can be kept warm.
As mentioned, the shinier the surface, the worse emitter radiation it will be. Therefore, the shiny teapots can be kept warm.
NEXT TIME WHEN YOU BECOME MOTHER PLS KNOW HOW TO BUY GOOD TEAPOTS =)

wah!!! the 2 teapots so sweet.. especially its my FAV PINK ! I WAN !!!
Actually, I like the 2nd one better..____________________________________________
VACUUM FLASK- 四全其美
It can keep liquid hot by minimising heat loss using- CCR and evaporation.
(not CPR pls..)
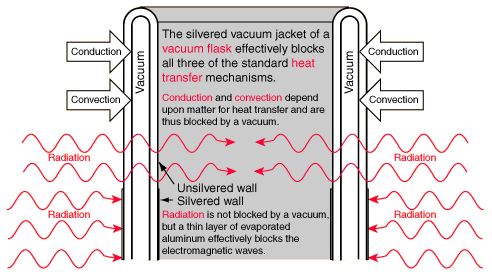
haha.. QWERTYUIOPAS}DWAFMLX")*)(#%
AS YOU KNOW
"PICTURE SPEAKS
A THOUSAND WORDS"
DBVXCNBMKXLAZXCVNBM
________________________________________
Chapter 8 for now (Intro for 8B, 8C & 8D)
Transferred process of thermal energy where it is transferred from hotter to colder region is by
- Conduction
- Convection
- Radiation
(3 processes in short form as CCR)
This video shows and educate you everything about CCR(in fact, everything about this chpt)
(notice the man draw the line so perfect)
Ok this is a clip taken in the past 1956. Though it is black and white, but its still interesting if u watch closely.. Some people may think Im weird but I find that old shows are nicer.. perhaps is the taste of the past..? I don't know .. like some shows 出路, 播音人, don't worry, be happy and 黄金路(new but old.. get what I mean?).. ok, but this is not a show. its an educative clip! Enjoy!
Sunday, August 31, 2008
Chapter 7
Solid, Liquid & Gas would be a short form as SLG
Water exists as SLG of 3 states
Lets start off with common basic in our lifes when we boil water

- Solid - the glass pot
- Liquid- the water inside
- Gas- steam
By the way, for your info, Fart can be considered a gas![]() . MUHAHAHA
. MUHAHAHA
VIDEO TIME !!!
-CHECK OUT this 2 website that may help you understand this chapter in a different perspective
ok..this website is made by some students.. and I guess its their schoolproject. Anyway, they are trying to explain the strong forces of attraction between particles. Their way of explaining is interesting & maybe to some people, you would find it easier to understand through this video.
This website is simple with animations.. quite cute though
- closely packed together and arranged in orderly manner
- large forces results results solid to have fix shape and volume
- It has high densities and vibrate in fixed position
*LIQUID
- clustered together & the space between molecules are wider as compared to solid
- Results in liquid have NO FIXED SHAPE
- free to move around in the boundary and has high density
GAS
- The molecules are very far apart & will occupy any space available, thus-low density
- This also explains why gas has NO fixed shaped or volume
- Travel in high speed
Solid has the strongest intermolecular force, followed by Liquid , then Gas
------------------------------------------------
- INSIDE SCOOP (interesting fact in animal life)
 As we make use of brownian motion's law, we know that as air rise. This rapid motion of air molecules help spread scent faster and hence on hot days, Emperor moths are able to pick scents faster in long distance.
As we make use of brownian motion's law, we know that as air rise. This rapid motion of air molecules help spread scent faster and hence on hot days, Emperor moths are able to pick scents faster in long distance.1)BEFORE

2)AFTER..tada !
 OK.. start now ! Lets make physics more interesting by watching simpsons- the truth is this movie only concerns a little on physics .. but its pretty funny !! I want to start off with saying that although I would try real hard to makes it as short as possible(I don't want this to be a dictionary).. But some concepts that are necessary would be present
OK.. start now ! Lets make physics more interesting by watching simpsons- the truth is this movie only concerns a little on physics .. but its pretty funny !! I want to start off with saying that although I would try real hard to makes it as short as possible(I don't want this to be a dictionary).. But some concepts that are necessary would be presentTesting..
Will start off with my favourite PINK
I must admit this is my 1st time in my entire life trying this out.
Must challenge myself as my leader always say :" lets tear down any walls within our minds. For its often we ourselves that determine our limits. "
- Daisaku Ikeda





